What is KERF?
How Much Kerf Waste
A large government lab asked the following:
"I was wondering if you knew much about the silicon wafer fabrication process. More specifically the kerf waste recovery methods silicon wafer manufactures implement. I have a project and we are trying to estimate the waste generated (around 40% for solar cell fabrication) but also account for waste recovery. There are a lot of waste recovery methods, but I don’t know what is commonly done in industry."
Please reference #270981 for our answer.
Get Your Quote FAST!
What Kerf Waste Recovery Methods Do Silicon Wafer Manufacturers Implement?
Whether you're new to the silicon wafer industry or if you're a long-time veteran, it's  important to know what kerf waste recovery methods silicon wafer manufactures implement. These methods are used to recover valuable material from the wastes produced when manufacturing silicon wafers. The methods discussed in this article include RF quasi-steady state photoconductance (QSSPC) method, Ramp settling tank, and Electrophoresis.
important to know what kerf waste recovery methods silicon wafer manufactures implement. These methods are used to recover valuable material from the wastes produced when manufacturing silicon wafers. The methods discussed in this article include RF quasi-steady state photoconductance (QSSPC) method, Ramp settling tank, and Electrophoresis.
RF quasi-steady state photoconductance (QSSPC) method
Using a contactless RF quasi-steady state photoconductance (QSSPC) method, a wide range of boron doped p-type Cz silicon wafers are measured for their photoconductance lifetimes. The effects of various chemical processes on the lifetime of the silicon is a key factor in the fabrication of silicon solar cells. Using the QSSPC method, one can obtain an immediate measure of the quality of the crystal growth on the wafer, which may serve as a guide in the future. Moreover, one can quantify the excess minority-carrier lifetime and the corresponding effect on the effective thickness of the material. The QSSPC method, which is not to be confused with the RF transient PCD method, is a good start in demonstrating the effects of light-induced carrier lifetime degradation.
The QSSPC method is best described as the alchemical equivalent of a thermal diffusion step, which would make sense in the manufacturing of PV cells. As a result, one can measure the effectiveness of a variety of boron doped p-type silicon wafers at different injection levels. Interestingly, the excess carrier density of a thin Cz-Si (111) wafer was correlated with the effective thickness of a thick sample exhibiting high surface recombination velocity. The SRV is typically in the order of 10-20 cm/s. It is not uncommon for an effective lifetime of over 200us to be attributed to the P-type bulk lifetime of the brick material.
The most important part of the QSSPC method is a thorough characterization of the inflection point of the sample. This can be attributed to the fact that a silicon wafer is detachable from each brick. The brick is subsequently induced to fracture along the ion stress layer. In addition to determining the effective thickness, the QSSPC method can be used to quantify the effects of light-induced carrier lifetime degradation. This is a necessary step in the fabrication of high-quality silicon solar cells. The QSSPC method may be applied to other new materials such as gallium arsenide, niobium, and tungsten. The resulting high-quality silicon solar cells should prove to be a boon for the silicon industry.
Electrophoresis
Using the kerf waste recovery method is a very efficient way to reduce the amount of waste. Kerf waste is generated from the wire cutting process used to cut polycrystalline silicon ingots. The waste material contains abrasive particles and lubricating liquid. These particles can be separated to remove metallic and non-metallic impurities. The use of the kerf as a feedstock may be more environmentally friendly than landfilling the sludge.
Several researches have been conducted to determine the removal efficiencies of metallic and non-metallic impurities from silicon kerf waste. Some studies have also investigated the recycling of sludge waste from diamond wires. In addition, the use of acetone as a lubricant has been studied. The efficiency of metal impurities removal was also investigated using acid solutions.
SiC and Si powders were found to be present in the kerf loss slurry. The particles were analyzed for their size, volume and number of particles. SiC powders had a larger particle size than the polished powder. The SiC phase was confirmed by X-ray diffraction (XRD) and electron diffraction spectroscopy (EDS).
Using the FE-SEM, the crystalline structures of the particles were observed. A spherical primary particle was observed with a diameter of about 10 nm. A number of SiC and Si particles were observed, with the majority of them being sharp corners. These particles were also observed alongside amorphous SiOx particles. The agglomerates of nanometer particles were around 300 nm in diameter.
A kerf waste recovery method is used to separate SiC and Si powders from sludge. The kerf waste recovery method is an inexpensive method to reduce the amount of waste. However, the process can be time-consuming and expensive.
Sludge waste generated during the slicing process is usually composed of abrasive particles and lubricating oil. The sludge contains Si and SDS. Adding SDS to the kerf waste recovery process has been reported to improve the separation of SiC and Si powders. However, it is important to note that adding SDS to the kerf waste recovery method may lead to reduced purity of SiC and Si powders in the MIBC phase.
Ramp settling tank
Traditionally, silicon carbide was used in slurry slicing techniques. The main concern for previous studies was the removal of SiC. In this study, we present a new method to achieve this goal: a ramp settling tank.
A ramp tank has two key features: it can capture tiny particles at the collection port and it has an improved separation performance compared to a standard rectangular tank. Both features increase the recovery rate of SiC powders.
The smallest particles captured by the ramp tank were around one mm in size. This smaller size reduced the time it took for particles to reach the bottom of the tank and therefore reduced the interference with settling. The amount of SiC powders purified by the ramp tank was significantly higher compared to the standard rectangular tank.
The kerf, which is composed of metallic contaminants, organics from a polymeric lubricant, and some moisture, had a pore volume of 0.052 cm3 g. The kerf also exhibited a BET of 0.5.
The kerf had a surface area of 9.6 m2 g. It also had the highest MW polymer of all the particles tested. The kerf contained 0.3% aluminum and 12% carbon as tar.
The kerf also provided some elemental analysis. A simple analysis using TGA showed that the weight loss of the kerf was modest.
The kerf was dry before experiments. A sample was collected at various positions in the tank. The kerf also supplied the elemental analysis that the tank exhibited.
The best separation performance came at position 10 in the ramp tank. The kerf was collected and cleaned to remove the moisture and metallic contaminants. It also provided the elemental analysis. The results were not as good as those obtained at position 8, where the kerf contained higher percentages of SiC. In the rectangular tank with baffle plates, the kerf exhibited a lower percentage of SiC.
The kerf also supplied the EDS spectrum of b. This is a more impressive feat than the smallest particle in the tank. The zeta potential of Si and SiC particles has an impact on their separation.
Chemical process
Various semiconductor and photovoltaic industries generate significant amounts of kerf silicon waste. This silicon powder waste can be recovered and converted into high purity silicon. This process requires a series of chemical and process steps. However, these steps are not enough to ensure high purity PV grade silicon. It is important to develop new technology for c-Si recovery from silicon grinding material.
Silicon powder waste can contain abrasives, metals, or processing oils. The final product is generally a slurry of silicon, abrasive, and carrier fluid. This slurry is mainly composed of silicon carbide and metal impurities. It has a particle size of up to 10 mm. The kerf silicon is typically purified through acid leaching.
The process to recover silicon from kerf silicon waste includes the separation of silicon carbide and metal impurities from the kerf silicon. In addition, chemical pretreatment can be applied to reduce metal content. Chemicals can include hydrofluoric acid, HCl, hydrochloric acid, hydrobromination, and hydrochlorination.
The final product of the acid leaching process is a condensed liquid of silicon-containing compounds. The process is performed at a temperature of 200-250 deg C. Hydrofluoric acid is used to reduce the oxidized silicon. Hydrochlorination is also a potential method to decrease the silicon oxide layer.
Physical pretreatment is also an option for silicon powder wastes. The surface impurities can be removed by physical processes such as washing or scrubbing. The surface impurities can be treated by slag treatment to diminish the silicon oxide layer.
The final kerf silicon waste is estimated to be a mixture of carrier fluid, metallic and non-metallic impurities. This kerf silicon waste may also be composed of polyethylene glycol. The waste is typically treated with high purity water.
The recovery process can be based on density or momentum. Density-based processes may include flotation, centrifugation, and momentum-based separation techniques. A column type scrubber can be utilized for this process. Magnetic strips may also be used in the wash column to remove iron oxide solid materials.
The authors are developing a factory project that would capture this opportunity and recover silicon. They also aim to develop a technology for the recovery of high purity c-Si from silicon grinding material.
Types of Kerf Waste Recovery
Choosing the best kerf waste recovery solution depends on the type of industry you are 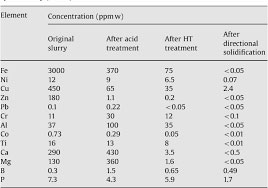 in, your goals, and your budget. Generally speaking, there are three types of kerf waste recovery: chemical kerf waste recovery, physical kerf waste recovery, and organic kerf waste recovery. Among these three types, Metallurgical grade silicon kerf waste recovery is the most common and widely used process, followed by acid extraction, pretreatment, and metallurgical reduction. Metallurgical grade silicon is often used as a component for semiconductors, while acid extraction and pretreatment are used to produce materials such as glass, ceramics, and cements.
in, your goals, and your budget. Generally speaking, there are three types of kerf waste recovery: chemical kerf waste recovery, physical kerf waste recovery, and organic kerf waste recovery. Among these three types, Metallurgical grade silicon kerf waste recovery is the most common and widely used process, followed by acid extraction, pretreatment, and metallurgical reduction. Metallurgical grade silicon is often used as a component for semiconductors, while acid extraction and pretreatment are used to produce materials such as glass, ceramics, and cements.
Metallurgical reduction process
Metallurgical reduction process for kerf waste recovery is a method to recover high purity metallurgical silicon from silicon kerf waste. The process converts a mixture of kerf material, silicon (Si) and abrasive reducing agent to high purity metallurgical silicon. Metallurgical silicon is used as a feedstock for polysilicon production. This high purity silicon is also suitable for other metallurgical processes such as metallurgical alloy production.
The process may be employed as an alternative to other silicon recovery processes, which typically require the removal of SiC. The kerf material can be treated to reduce silicon oxide to silicon. In addition, metallic impurities can be reduced by acid mix. The kerf material mix can be further treated to consume the reducing agent. This process can be used to recover silicon from solar panels and other high purity silicon waste.
The first process involves in-situ formation of S1O2 in proportion to the silicon content of the kerf waste. The SiC content of the kerf waste can be added to the silicon content in the process. The final kerf silicon residue will have less metallic impurities and higher purity.
The metallurgical reduction process can be performed at temperatures ranging from 1500C to 2000C. The leaching efficiencies vary from 80 % to 80%. The process may be performed on lumpy quartz or Si metal. It may also include a pretreatment wash. The final kerf silicon residue will be a mixture of SiC and carrier wire. The residue is estimated to contain traces of metals from finishing stages. It will also have an estimated carrier fluid.
The total residue from the Oxidized Si process was analyzed for Fe and alkali metals and the levels were less than 10 ppm and less than 20 ppm, respectively. The levels of Cu, Zn, and Al were between 20 and 500 ppm.
The second process includes the addition of high purity Si02 to the kerf material mixture. This is a substitute for oxidizing silicon. The Si02 can also be added to intrinsically pure silicon. It is estimated that the level of Si02 is equal to the content of SiC.
Acid extraction
Typically, silicon kerf waste is composed of a 35% SiC content and 5% Fe content. In addition, it is contaminated with trace metallic impurities and nonmetallic impurities. Silicon kerf waste is used in PV feedstock industries and metallurgical processes.
During the kerf recovery process, the metallic and nonmetallic impurities are removed by acid washing. The slurry is then purified and converted to metallurgical silicon. The process also uses superficial oxides and a submerged arc furnace. The final kerf silicon residue contains trace amounts of metals from the finishing stages of the slurry recovery operation.
In addition, silicon impurities, such as Al, CnHmOl, and SiC, are present as oxides. The presence of these impurities in a kerf silicon mixture can determine the degree of the carbon oxidation.
In order to determine the amount of carbon eliminated from the kerf, three thermogravimetric analysis methods were used. These methods were based on the analysis of the BET, OFW, and KAS peaks. These three instruments were used in conjunction with each other, which provided a more precise determination of activation energy.
A total of ten peaks were identified. Peak identification is based on the NIST library. Peaks were assigned to references and integrated using the Agilent Mass Hunter Quantitative Analysis Software. The error bars represent standard deviation.
The presence of Al in the kerf was found to be only 0.3%. The peak at 1612 cm-1 is due to vibration of Al2-O3 and originates from the beam supporting the ingot during the slicing process. The peak in air and N2 are similar.
Similarly, the peak at 1612 cm-1 in the N2 case is characterized by a similar amplitude to that in air. The MW of the polymer is higher in the N2 case.
These results show that a greater percentage of carbonaceous compounds are removed from the kerf in the N2 atmosphere than in the air. However, 12% of carbon remains in the kerf as tar.
In addition, a broader range of metal impurities are present in the kerf than in the air. In particular, aluminum is the third most abundant contaminant.
Pretreatment process
During the manufacturing of silicon wafers, the waste material is generated, known as kerf silicon waste. This material contains silicon, silicon oxide, and abrasive reducing agents such as silicon carbide. Kerf silicon waste can be used for metallurgical applications. Kerf-derived silicon is used in the manufacture of silicon carbonitride and similar ceramic materials. This material is typically very pure, with low boron and trace metallic impurities.
A direct silicon recovery process is an environmentally friendly method of separating and purifying kerf silicon waste. It removes metals, non-metallic impurities, and oils from the carrier fluid of the waste. The resulting silicon is high purity and usable in metallurgical processes. The process also eliminates the need for environmentally deleterious kerf silicon disposal.
The process is designed to convert kerf material to a metallurgical grade silicon with a carbon content less than 100 ppm. This material can be used as a feedstock for polysilicon manufacturing. It can also be used in the manufacture of silicon oxycarbide. The process is used for metallurgical grade silicon as well as for solar grade silicon.
The kerf-derived silicon is made through an electric arc furnace process. It is then cut into a wafer. The process may be used with other wire saw processes. The resulting silicon has a boron partition coefficient of 0.8. It can then be used as a substitute for MG-Si. This process also removes the minor extrinsic metallic impurities.
The method of separating kerf silicon waste can include washing with a high purity water. Other methods include chemical and physical pretreatment. It may also include reducing the magnetic content of the waste. The process may also include reducing the magnetic content of the carrier fluid.
The method of separating kerf silicon waste also may include the use of an organic-based liquid extractant. This type of extractant will help remove oils from oil-based kerf silicon waste. The process can also include chemical processing to reduce the metallic content.
In the process of separating kerf silicon waste, a number of pretreatment washes may be performed to remove extrinsic metallic impurities. It may also include multiple acid leach treatments. The amount of acid used is dependent on the amount of metals present in the kerf.
Metallurgical grade silicon
Metallurgical grade silicon is silicon with less than one part per million of metal impurities. This material can be obtained from kerf waste. The recovered material can be used in the manufacturing of silicon carbonitride, silicon oxycarbide and similar ceramic materials. Metallurgical grade silicon can also be used as silicon feedstock in the manufacture of polysilicon.
The present invention relates to a process for the recovery of kerf silicon waste. The waste is produced during the manufacture of silicon wafers. The waste is generally contaminated with water, non-metallic impurities and trace metallic impurities.
The waste can be recovered using physical or chemical methods. Physical methods include using selective laser energies and wavelengths to densify the silicon powder. Physical methods also can be applied to surface impurities. These processes will typically have particle sizes in the range of 1-10 microns.
Chemical methods can also be applied to silicon powder wastes. In addition, the silicon powder waste can be converted to a gaseous silicon intermediate. This intermediate can be further purified through distillation.
The present invention relates to the recovery of high purity Metallurgical grade silicon from kerf waste. The kerf material mixture is then converted into Metallurgical grade silicon by a metallurgical reduction process. This process is performed in an electric arc furnace. It is performed in the temperature range of 1500C to 2000C.
The process can be applied to kerf wastes that have metals or abrasives in them. The metal content of pretreated kerf silicon is usually one part per million (ppm) of the metal, such as copper or iron. The abrasive in the silicon waste is typically diamond abrasive.
The kerf waste can be pretreated to remove the metallic and non-metallic impurities. In order to achieve high purity, the Si0 must be added to the waste at a ratio of at least 99%. The silicon is then reacted with hydrogen chloride, silicon tetrachloride, and/or hydrogen gas to form the gaseous silicon intermediate. The resulting silicon is then purified by distillation.
Metallurgical grade silicon is then used as silicon feedstock in the manufacture of metallurgical alloys and polysilicon. The recovery process enables the environmental disposal of kerf silicon waste without generating any further waste.
