What is a Semiconductor Wafer?
A semiconductor wafer is a thin slice of material, typically made of silicon, that is used in the manufacturing of electronic components such as integrated circuits (ICs), microchips, and other semiconductor devices. The wafers are circular in shape and range in size from a few millimeters to several inches in diameter.
The wafer serves as the substrate for the electronic components, and it undergoes a series of complex manufacturing processes, including photolithography, etching, and doping, to create the necessary structures and electrical properties required for the specific application. The patterned layers of materials on the wafer form the integrated circuit, which can contain millions or even billions of transistors and other components.
Semiconductor wafers are a critical component in the electronics industry, and the size and quality of the wafer can impact the performance and efficiency of the resulting electronic devices. They are typically manufactured in specialized cleanroom environments using advanced equipment and processes to ensure high-quality and reliable products.
Semiconductor Wafer List
Semiconductor wafers can be made from various materials, but the most commonly used substrate is silicon. Other substrates that are considered semiconductor wafers include:
- Gallium Arsenide (GaAs)
- Silicon Germanium (SiGe)
- Indium Phosphide (InP)
- Sapphire (Al2O3)
- Silicon Carbide (SiC)
- Gallium Nitride (GaN)
- Zinc Oxide (ZnO)
- Diamond
These substrates have different electrical and mechanical properties, making them suitable for various applications in the electronics industry, such as high-frequency devices, power electronics, optoelectronics, and more. However, silicon is the most widely used substrate due to its abundance, cost-effectiveness, and well-established manufacturing processes.
What is a Semiconductor in Layman's Terms?
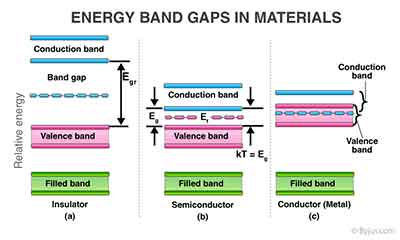 The semiconductor is a type of material that consists of atoms that are arranged in a three-dimensional periodic structure. One type of semiconductor is pure silicon, which contains negligible amounts of impurities. In this crystal, a silicon atom has four electrons in its outer orbit, surrounded by four nearest neighbors. These atoms share electron pairs, which form covalent bonds. When two atoms are in close proximity, their ions are attracted to one another.
The semiconductor is a type of material that consists of atoms that are arranged in a three-dimensional periodic structure. One type of semiconductor is pure silicon, which contains negligible amounts of impurities. In this crystal, a silicon atom has four electrons in its outer orbit, surrounded by four nearest neighbors. These atoms share electron pairs, which form covalent bonds. When two atoms are in close proximity, their ions are attracted to one another.
Semiconductors are a combination of insulators and conductors. In particular, a semiconductor is an insulator and is used to conduct electricity. However, its conductivity depends on two factors: the control electrode and the intensity of irradiation. Many of today's electronic products are made of semiconductors, from smartphones to appliances and medical equipment. If you've been wondering what a semiconductor is, here's a quick primer.
A semiconductor is an electronic material made up of two materials, an insulator and a conductor. They allow current to flow in one direction, but not the other. They are used to convert AC signals to DC, and allow the positive part of the wave to pass through. This makes them the most basic of all semiconductor devices. It's not just the electronic devices that use them. You'll find them in appliances, video games, medical equipment, and more.
A semiconductor is made of several different types of materials. A semiconductor is a material that is neither an insulator nor a conductor. Its characteristics make it an ideal medium for controlling electrical current. A semiconductor can be either an insulator or a conductor. A superconductor has no resistance, allowing electrical current to flow forever in a closed loop. These materials are used in electronics and in computer memory.
A semiconductor is a device that allows current to flow in one direction. A diode is a single-channel electronic device, which means that it allows only one-directional current to flow. For this reason, it's also a very useful device for converting an AC signal to a DC. The simplest semiconductor is a diode, which functions like a one-way turnstile for electrons.
A semiconductor is a material that can be either a conductor or an insulator. Its conductivity depends on a control electrode and the intensity of an irradiation. A semiconductor can be a conductor. It can also be an insulator. The term semiconductor is an electronic material that is used in various devices. When compared to insulators, it is an electrical conductor.
A semiconductor is a type of solid that has specific electrical properties. It is the foundation for computers and other electronic devices. In addition to electronics, a semiconductor is usually a solid chemical element. It can conduct electricity under certain conditions and is therefore the perfect medium for controlling electrical current. There are two types of semiconductors: a n-type semiconductor is a pure insulator. The other type is a semiconductor.
A semiconductor is a solid material that has specific electrical properties. It is often a solid chemical element that can conduct electricity under certain conditions. Because of this, it is an ideal medium for regulating electrical current. Other types of materials can be classified as insulators and conductors. A semiconductor sits between the two. For example, a diode is a semiconductor. A transistor is a semiconductive element.
A semiconductor is a material with electrical properties that lie between those of a conductor and an insulator. Its properties are intermediate between a conductor and an insulator, and they have a high degree of mobility. A semiconductor is an ideal medium for controlling the flow of electrical current. A semiconductor has low thermal and electrical characteristics. In contrast, an insulator is a nonconductor.
c-Si or Crystalline Silicon are used in Semiconductors. Using several micro-fabrication processes, an Ingot of Silicon is turned into a thin Silicon wafer.
The Processes Include:
- Doping
- Ion Implantation
- Etching
- Thin-Film Deposition
- photolithographic patterning
Microcircuits are etched onto the wafer. They are then separated dicing, then the diced pieces are packaged as an integrated circuit.
Here are some facts you need to know about semiconductor wafers:
1. Semiconductors are made from Silicon mined from beached. Silicon is the 2nd most common element on earth, after carbon-which is what we are mde of!
2. Only a few beaches can be mined for Silicon. One in Australia and one in Italy. Pristine sand is required.
3. Semiconductor wafers are available in a variety of diameters. In 1960 the first semiconductor wafer was manufactured in the US, the semiconductor wafer’s diameters was 1 inch. Today the standard semiconductor wafer size is 12 inch with foreseeable plan to achieve 18 inch semiconductor wafers.
4. Silicon ingots are grown from a "seed" almost like a plant, but in a cylindrical tube.
5. There are two processes that can be used. One method is called Czochralski process the other is Float Zone. The difference of the two methods are in their use.
6. Semiconductor wafers thickness can be as thin as 2 micron! Your hair is 100 microns.
What is The effect of Temperature on a Semiconductors?
A semiconductor device works by controlling the movement of charge carriers, which move in a certain direction. It does so by arranging them in layers with different electrical properties. In this way, it is possible to design circuits that operate by moving charge carriers. One example of a semiconductor is the p-n junction, which favors carrier movement in one direction and hinders it in the opposite direction. As a result, the electrical difference between the n and p regions disappears at sufficiently high temperatures.
A semiconductor changes in electrical conductivity according to the temperature. As the temperature  increases, the kinetic energy of electrons in the crystalline solid also increases. At room temperature, the atoms only move around in a single direction: they are vibrating around a stable lattice position. At higher temperatures, atomic motion is limited to vibration of the lattice, resulting in greater electrical resistance.
increases, the kinetic energy of electrons in the crystalline solid also increases. At room temperature, the atoms only move around in a single direction: they are vibrating around a stable lattice position. At higher temperatures, atomic motion is limited to vibration of the lattice, resulting in greater electrical resistance.
The increase in temperature decreases the resistivity of the semiconductor. As the temperature increases, the electrons lose their covalent bonds, and the semiconductor becomes an insulator. When the temperature increases, the free electrons in the conduction band jump to the valence band and the electrical conductivity increases. This makes the semiconductor a conductor. The increase in electrical conductivity is the effect of thermal energy, which sets free electrons in the conduction band.
The energy-band diagrams of the semiconductors are shown in Figure 12.6.3. Silicon and Germanium have a small band gap, while diamond has a wide band gap. The higher the temperature, the smaller the band gap and the larger the electrical conductivity. Therefore, the effect of temperature on a semiconductor is a fast increase in electrical conductivity. This is because the thermal energy is needed to break the covalent bonds and free electrons in the semiconductor.
The temperature also affects mobility. When an electrical conductor is heated, the charge carrier mobility decreases. This decreases the electrical resistance. It makes it more suitable for high-voltage applications. Heat will cause the conduction band of a semiconductor to increase. As the heat is applied, the thermal energy is absorbed by the semiconductor. Moreover, the hole is the center of attraction for an electron and is a source of electricity.
A semiconductor has two bands. The valence band is filled with electrons. The conduction band is empty. When the valence band is filled with electrons, it will have high electrical conductivity. A large gap, on the other hand, will make a semiconductor an electrical insulator. The third type of semiconductor is the electrical insulator. The conductive band is the upper band.
As the temperature increases, the electrons in the semiconductor will decrease their mobility. In other words, their electrical conductivity will increase. A partially filled band is an electrical insulator. At the same time, a semiconductor is an insulator. The lower band is an electrically conductive material. At the same time, the valence band is an electrically insulator.
The temperature increases the electrical conductivity of a semiconductor. At low temperatures, a semiconductor is an insulator, and it increases the electrically conductive band at higher temperatures. The higher the temperature, the less electrically-conductive the semiconductor. In contrast, a partially filled band causes a reduced electrical conductivity. However, the lower band is an electrically insulator.
In a semiconductor, an electrical field decreases the intrinsic mobility. The more atoms in the lower temperature, the higher the intrinsic mobility. A semiconductor with high temperatures will experience more electrons. An increased temperature decreases the intrinsic mobility. Consequently, the device will experience a decrease in electricity. This can result in a higher temperature. A higher temperature decreases electrical resistance.
What is the Variation in Semiconductor Parameters with Temperature?
Watch the video!
What is Extrinsic Semiconductor?
When we talk about semiconductors, the term 'extrinsic' has various meanings. The term extrinsic means that it is not intrinsically made of silicon. This term comes from the French language, and it is an alternative name for extrinsic. In this article, we will discuss the differences between extrinsic and intrinsic semiconductors. In addition to their basic differences, we will also touch on their doping, electrical neutrality, and Electron acceptor dopants.
Doping
Doping in extrinsic semiconductors is a process that adds an extra valence electron to one or more atoms to make the compound more semiconductor-like. In semiconductors, dopants can be an ion or a molecule. The doping atoms are commonly metals, such as boron and sulphur. In many cases, the extra valence electron is not needed, as the substance is electrically neutral.
Pure semiconductors are made by doping them with pentavalent elements, such as boron or lanthanum. The pentavalent element forms a covalent bond with the four electrons of the parent atom. The pentavalent impurity electron remains weakly attached to its parent atom, enabling it to move about the crystal lattice with minimal ionisation energy.
A large gap between the valence and conduction bands of a semiconductor is a sign of an insulator. Metallic samples, by contrast, have overlapping bands, making them excellent conductors of electricity. When the semiconductor band gap becomes narrower, electrons can jump from one band to the other, increasing the conductivity of the material. The difference between the two band gaps is due to the amount of impurity added to it. This process is called doping, and involves careful control over the impurity types.
Electrical conductivity
The majority carrier in extrinsic semiconductors undergoes various phases when heated, with the amount of dopants remaining constant. In n-type semiconductors, the majority carrier begins in the Freeze-Out Region, where free electrons cannot move. At higher temperatures, they enter the blue regime, where donor electrons are promoted into the conduction band. During this period, the overall mobility of electrons decreases, as impurities scatter.
Extrinsic semiconductors have different levels of conductivity depending on their dopant content. In a n-type semiconductor, an extra electron is added, and this electron is of a higher energy level than its counterpart in silicon. This extra electron jumps across the small energy gap, which is only 1.1 eV in a pure semiconductor. The concentration of holes in an extrinsic semiconductor depends on the amount of dopant.
Electrical neutrality
An electrically neutral semiconductor is a material in which the charge of a donor ion is the same as that of the electron of its host atom. This property is referred to as extrinsic semiconductor. It can be either a p-type or an n-type. An n-type semiconductor contains extra electrons that can complete the covalent bonds in its crystal. A p-type semiconductor contains a lone electron on the host atom.
The mobility and conductivity of extrinsic semiconductors depends on the relative concentration of the electrons and holes in the material. In p-type semiconductors, the total electron concentration is equal to the number of holes while the electrons are less than half of the donor sites. The difference in the amount of conduction electrons and holes is the measure of the carrier concentration. This makes n-type semiconductors more conductive than p-type semiconductors.
Electron acceptor dopants
One way to increase the amount of conduction in extrinsic semiconductors is by adding an electron acceptor dopant. Such a substance contains four outer electrons which combine with the silicon atom to create the material's intrinsic conductivity. A fifth electron, on the other hand, is free to move and serves as a charge carrier. Free electrons require less energy to transfer from the valence band to the conduction band.
While conducting at room temperature, intrinsic semiconductors have low conductivity. This low conductivity makes them unsuitable for electronic devices. To address this problem, the concept of doping was developed. Extrinsic semiconductors are semiconductors that have been doped with specific impurities in order to change their electrical properties and make them more usable for electronic devices. Here's an example of how this process works.
Video: Extrinsic Semiconductor Explained
What is Intrinsic Semiconductors?
You may have heard about semiconductors before but if you're not familiar with them, then you're missing out on a lot of great information. Intrinsic semiconductors are a kind of material that behaves like an insulator at room temperature but turns into a semiconductor when heated. When this happens, electrons move from the valence band to the conduction band and leave an equal number of holes.
Conduction
Intrinsic semiconductors are solid-state materials that conduct electricity at low temperatures. At room 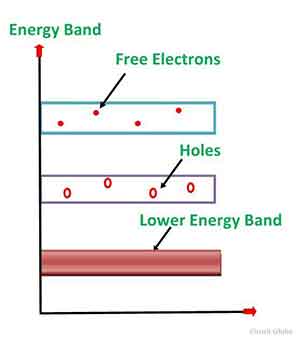 temperature, electrons in the conduction band are free, and other electrons move to fill the vacancies left by free electrons. These vacancies are known as holes, and they contribute to the conductive properties of the intrinsic semiconductor material. The density of energy states in the semiconductor affects the amount of electrons in the conduction band, and temperature is a crucial factor.
temperature, electrons in the conduction band are free, and other electrons move to fill the vacancies left by free electrons. These vacancies are known as holes, and they contribute to the conductive properties of the intrinsic semiconductor material. The density of energy states in the semiconductor affects the amount of electrons in the conduction band, and temperature is a crucial factor.
When an electric field is applied, electron-hole pairs can drift. This means that the electrons move in opposite directions, and the holes move in the direction of the electric field. As a result, the movement of electrons and holes is reflected in a conductive circuit. This phenomenon is called a band gap, and it describes how the conduction of a semiconductor is achieved. In a typical semiconductor, there are an equal number of holes and electrons.
Electrons
The density of carriers in an intrinsic semiconductor is determined by the concentration of valence electrons. The density of carriers varies with temperature. When the temperature of an intrinsic semiconductor is 300K, the density of carriers is approximately 1010 cm-3. In addition, intrinsic semiconductors are doped in one or both directions. These doped materials are still intrinsic semiconductors. However, they are doped with both acceptors and donors equally.
The electrons in an intrinsic semiconductor behave like an insulator at room temperature. When the temperature increases, the electrons in the outermost orbit break the covalent bond with the atom's nucleus and jump into the conduction band. In the conduction band, the electrons do not have a nucleus and are free to move. A hole is created in the valence band when an electron in the outermost orbit moves out of this orbit into the conduction band.
Hole movement
The motion of holes and electrons in a semiconductor is what drives the conductor current. Electrons in the conduction band move towards the anode of the semiconductor while holes move towards the cathode. This process continues in a cycle mode. Ultimately, the electrons and holes are paired and the current flows. However, in many semiconductors, the holes and electrons do not move in the same direction.
The difference between doped and intrinsic semiconductors is the source of electron holes. Holes in an intrinsic semiconductor are formed when electrons are thermally excited. In contrast, doped semiconductors have holes supplied by impurities. Pure silicon contains no impurities that can move electrons. Therefore, electrons move across the material when they are excited by an external voltage. This process is referred to as hole movement.
Band gap energy
The band gap energy is a fundamental property of intrinsic semiconductors. This measure describes the minimum change in energy required to excite an electron. In order for electrons to participate in conduction, they must be excited. Therefore, the band gap energy of a semiconductor varies depending on its composition. The band gap energy of an intrinsic semiconductor is often measured using atomic force microscopy. The following table describes the band gap energy of a semiconductor.
In addition to the band gap energy, the semiconductor's conductivity increases exponentially with temperature. Thermally generated carriers are especially useful for measuring temperature. The energy gap is the difference between the energies of the highest conduction band and lowest valence band levels. Depending on the type of semiconductor, the band gap energy is different from the one in the valence band. This property is called intrinsic carrier concentration. Hence, the intrinsic carrier concentration is equal to the number of broken bonds in an intrinsic semiconductor.
Processes used to make Intrinsic Semiconductors
The basic concept behind intrinsic semiconductors is that their structure is made up of two layers connected by covalent bonds. The energy released during the recombination of electrons and holes is converted into electromagnetic radiation and thermal vibration of the lattice. At room temperature, only a few electrons can break the covalent bond, leaving an empty position known as an electron hole. When a voltage is applied to both sides of an intrinsic semiconductor, the free electrons move in a directional manner, filling the electron holes along the way. These free electrons are moving in the opposite direction to the electrical current, thus allowing the device to operate in an inductive manner.
An intrinsic semiconductor can be classified as either an N-type or a P-type semiconductor, depending on whether it is doped or not. The former is characterized by a pentavalent atom of Antimony, while the latter shows a trivalent atom of Boron that forms a void space within the crystal structure that attracts electrons. However, the former is a better semiconductor choice because it has no impurities on the crystal structure, allowing for an equal number of holes and electrons.
Video: Intrinsic Semiconductors Explained
What is a Light Absorbing Semiconductor?
Light absorbed by a semiconductor occurs when photons have an energy higher than the band gap. The electrons in the conduction band fall back to the bottom of the semiconductor, releasing the excess energy as heat. This process reduces the efficiency of the device. In contrast, light emitted by a semiconductor does not have this effect. Instead, it can cause the semiconductor to become transparent to light of the same wavelength. If you want to understand the process, you can view a semiconductor animation.
A light-absorbing semiconductor can also be a photocatalyst for fuel cells. The basic requirements for such a catalyst include a stable band structure, the ability to absorb sunlight in the visible light spectrum, and the ability to separate the charge of a solid and a liquid. Once this is accomplished, a semiconductor photocatalyst will be useful in converting CO2 and water. Further, this type of semiconductor photocatalyst will enable solar cells to generate electricity.
Another challenge in the development of semiconductors is the ability of these materials to convert light into heat. One way to overcome this is to design them for more energy efficiency. In order to develop light-absorbing semiconductors, researchers at the University of Buffalo have developed a strategy that will improve light absorption in these materials. In future, this research may be applied to nano-scale photovoltaics and camera sensors. But before they can produce these devices, they must focus on converting heat and light into energy.
Video: Optical Absoprtion in Semiconductors

 The semiconductor is a type of material that consists of atoms that are arranged in a three-dimensional periodic structure. One type of semiconductor is pure silicon, which contains negligible amounts of impurities. In this crystal, a silicon atom has four electrons in its outer orbit, surrounded by four nearest neighbors. These atoms share electron pairs, which form covalent bonds. When two atoms are in close proximity, their ions are attracted to one another.
The semiconductor is a type of material that consists of atoms that are arranged in a three-dimensional periodic structure. One type of semiconductor is pure silicon, which contains negligible amounts of impurities. In this crystal, a silicon atom has four electrons in its outer orbit, surrounded by four nearest neighbors. These atoms share electron pairs, which form covalent bonds. When two atoms are in close proximity, their ions are attracted to one another. increases, the kinetic energy of electrons in the crystalline solid also increases. At room temperature, the atoms only move around in a single direction: they are vibrating around a stable lattice position. At higher temperatures, atomic motion is limited to vibration of the lattice, resulting in greater electrical resistance.
increases, the kinetic energy of electrons in the crystalline solid also increases. At room temperature, the atoms only move around in a single direction: they are vibrating around a stable lattice position. At higher temperatures, atomic motion is limited to vibration of the lattice, resulting in greater electrical resistance.